 
We know that the solid form of water is ice and that the gaseous state is steam.įor water to become solid, it needs to be kept below 0 oC (at atmospheric pressure), which slows down the vibration of each of the molecules of the water until bonds are able to be formed between the water molecules. Conservative Forces and Potential EnergyĪ useful example for explaining the particle model of matter is water we use it in our day-to-day lives and have likely seen each of its states and how it changes between each of these states.Total Internal Reflection in Optical Fibre.They are close together and vibrate in position but don't move past one another. In a solid, the particles are very attracted to each other. Solids, liquids, and gases are made of tiny particles called atoms and molecules. Newton’s and Huygens’ Theories of Light Matter on Earth is in the form of solid, liquid, or gas.Einstein's Theory of Special Relativity.Centripetal Acceleration and Centripetal Force.Connecting Linear and Rotational Motion.Electromagnetic Radiation and Quantum Phenomena.Galileo's Leaning Tower of Pisa Experiment There are various characteristics of particles of matter that we will address further down.What are the characteristics of Particles of Matter. are all different kinds of matter because they occupy space, have volume and mass. Mechanical Energy in Simple Harmonic Motion Like sand, sugar, hydrogen, oxygen, air, water, petrol etc.Kinetic Energy in Simple Harmonic Motion.Displacement, Time and Average Velocity.Magnetic Flux and Magnetic Flux Linkage. 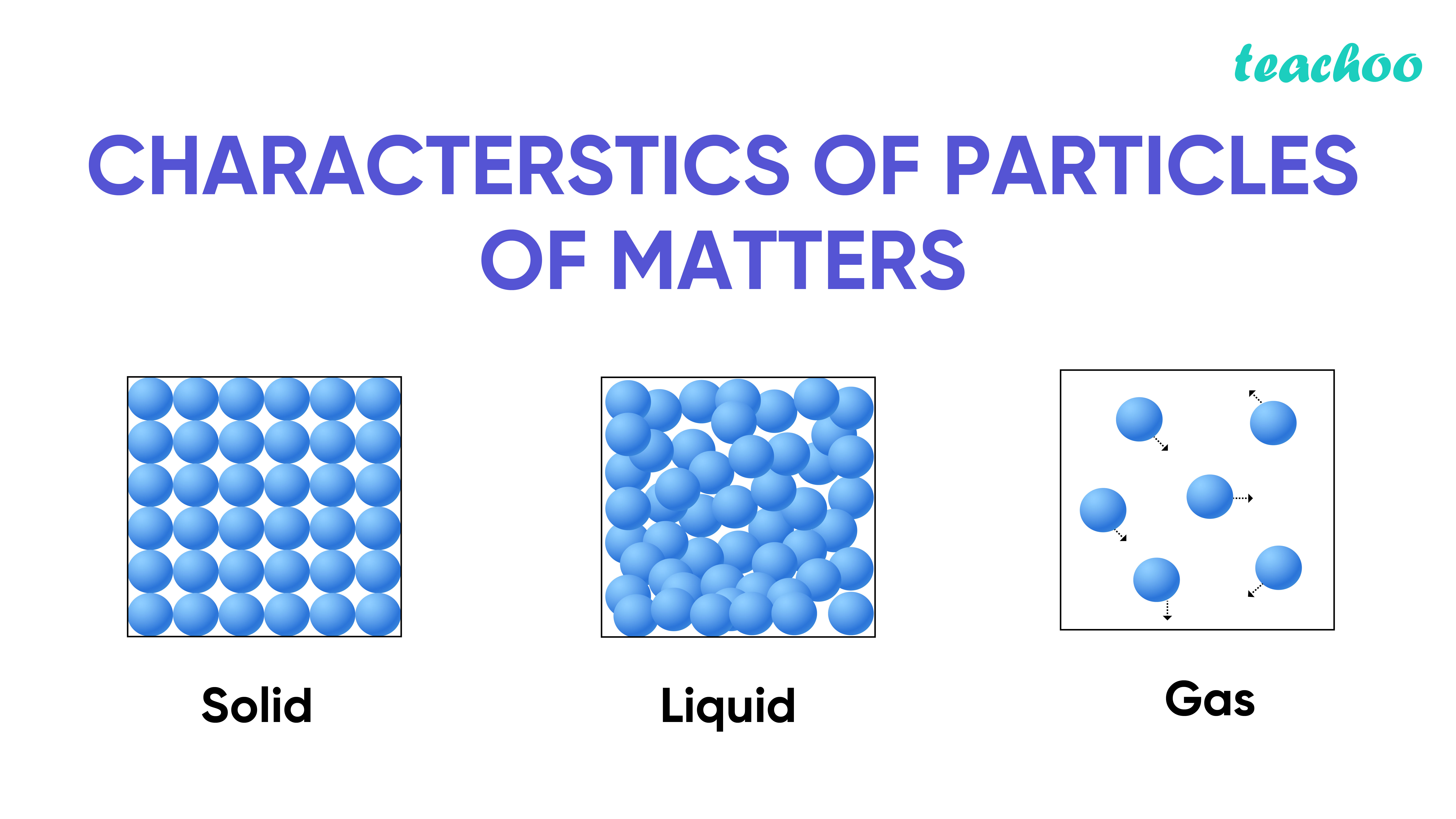
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
